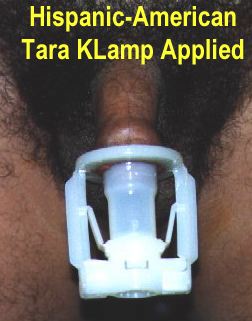
This is a Malaysian invention, developed by Dr. Gurchran Singh Tara Singh during the 1980s and early 1990s. It was first marketed in 1995. Gurch, as he is known to his friends, should properly be regarded as the father of the modern disposable circumcision clamp. His circumcision device is highly regarded in SE Asia and in Africa it is making inroads into the sub-Saharan market. In June 2009, Dr. Singh’s tireless work promoting safe circumcision earned him the Malaysian Medical Association’s Outstanding Public and Healthcare Services Award, a rare honour equivalent to a Lifetime Achievement Award.
As is so often the case with clever inventions, a host of similar devices soon followed in the wake of the original. Each one differs slightly, in an attempt to skate around the intellectual property rights of their competitors. Elsewhere on this website you will find descriptions of (in alphabetical order) the Ali’sKlamp, Ismail Clamp, KirveKlamp, SmartKlamp and Sunathrone devices.
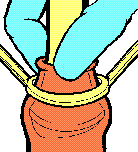
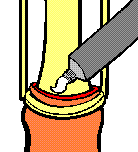
The principle of operation of the Tara KLamp is illustrated by the following diagrams, based upon the manufacturer’s original publicity material dating from the 1990s:
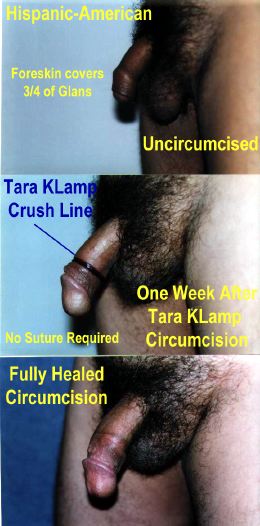
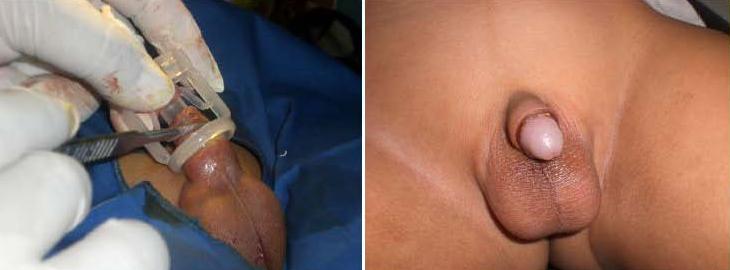
Once the clamp is closed, the fate of the foreskin is sealed. There’s no going back; the powerful crushing action causes irrecoverable tissue damage. In theory an option exists not to cut with a scalpel, instead leaving the condemned part of the foreskin in place to necrotise - the clamping action having totally cut off the blood supply. However, this approach is not recommended due to the possibility of the considerable quantity of dead tissue harbouring infection during the week or so until the clamp drops off naturally. Normally, a scalpel will be used to sever the foreskin a few minutes after the clamp is applied. The correct position of the cut is 2mm to 3mm clear of the distal face of the clamping ring. When marking out a circumcision using one of these clamps, it is vital to realise that the eventual scar line will form at the position of the proximal face of the clamping ring, not at the position of the scalpel cut.
Unlike the Plastibell device, which uses string to strangulate the condemned tissue, a Tara KLamp can achieve fairly tight styles of circumcision without the inner part digging into retained tissue.
The inventor claims the following benefits viz-a-viz conventional circumcision surgery:
- The device is delivered pre-sterilised.
- The latching mechanism is designed to ensure single use only, thus guarding against cross-infections (hepatitis, HIV).
- The device totally shields the glans from the scalpel, making the procedure very safe.
- The method is non-invasive; no sutures, ligatures or dressings are required.
- Haemostasis is maintained during surgery.
- There is no open wound.
- Results are predictably neat and even across a wide range of style choices.
- A sterile environment is not required. The device can be used in homes, schools, at religious sites and other non-medical places.
- There is no post-surgical bleeding or oozing. The patient can resume normal activities straight away.
In the years since the Tara KLamp was first marketed, a number of incremental design improvements have been made. Most notable of these is that the material used to mould the clamp has been changed. This has enabled the size of the device to be reduced without compromising on strength. Only clamps made the newer material should now be in circulation, any unused devices moulded using the old material now being past their use-by date in terms of sterility.

Sterilisation is by the EO process. The current grade of plastic used is not suited to sterilisation by Gamma-Ray irradiation (Cobalt 60 exposure). In common with many plastics, it goes brittle when irradiated. So, despite the environmental concerns that some people attach to the Ethylene Oxide process, the old-fashioned method prevails. Experimentation with sterilisation by ultra-high voltage electrostatic fields is in prospect, but not yet implemented.
Current clamp production is in a range of sizes from newborn to adult. From the outset, the production and marketing company TaraMedic supplied these clamps as part of a kit that also included a scalpel blade and a measuring device. This accords with the company’s original design concept, which envisaged a device useable in remote places by those with only ad-hoc training. Although a greater degree of medical expertise is now recommended, the concept of a "Field Kit" has been retained. No means of anaesthesia is included; users must organise that separately. The recommended form of anaesthesia is a Dorsal Penile Nerve Block (DPNB). For children, this can usefully be preceed by an application of a topical anaesthetic cream to dull the pain of the first injection.
On the subject of pain relief, Dr. Singh himself writes: “On discomfort, I would feel there could be some discomfort as the patient will have the device on him for a minimum of 4-5 days. However, we get feed-back that patients do not mind that, as they realize that the many benefits that they otherwise obtain, have really taken away their fears etc. and are grateful for that.”
The Tara Klamp seems to be most used currently in South Africa where
there have been some serious adverse outcomes reported. For details click
here.
Sources of further information about Tara KLamp
The manufacturer does not maintain a website at its Malaysian headquarters, but the South African branch does have a
site. Below are the contact details for supply enquiries. Note that TaraMedic will only make clamps available to qualified medical practitioners or properly constituted medical supplies wholesalers. Despite its original design concept as a device for use by "Barefoot Doctors" practical experience has shown that this NOT the case (see above) and it should only be used by qualified medical practitioners. The Tara Klamp is
most definitely not suitable for "DIY" circumcision.
TaraMedic Corporation Sdn Bhd
Technology Park Malaysia
Pusat Inkubator 3, Building 6,
1-3a Lebuhraya Puchong-Sg. Besi,
Bukit Jalil,
57000 KUALA LUMPUR
Malaysia
Tel: +603 89963667 and +603 89963668
Fax: +603 89963669